FTC disclaimer: This post may contains affiliate links and we will be compensated if you click on a link and make a purchase.
One of the biggest obstacles to good health is the occurrence of “free radicals” in your body.
Free radical damage is unavoidable— environmental pollution, high-fat diets, stress, and even sunlight contribute to free radical production.
Furthermore, your body generates free radicals during normal physiological processes in which your body organs, tissue, cell, and biomolecules work together to accomplish the complex goal of sustaining life.
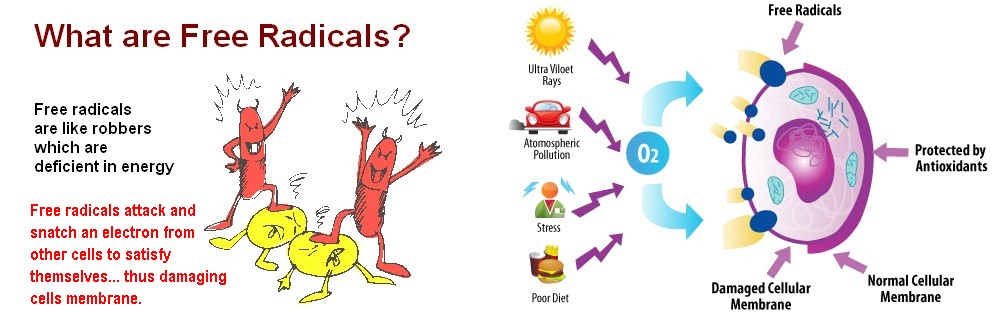
What are Free Radicals?
A free radical is an atom that has an odd number of electrons in its outer ring. In other words, it is imbalanced chemically.
Electrons are part of an atom. They are located at the outer shell of an atom. An atom usually becomes “grounded” or stable when each of the electrons pairs up.
The existence of an alone or unpaired electron is unnatural and makes an atom a free radical.
Due to this chemical imbalance, these unstable molecules (oxidants) are produced when your body breaks down food or environmental exposure to tobacco smoke, radiation, pollution, stress, and herbicides.
It is partially responsible for aging, tissue damage, and some diseases.
What is the problem with Free Radicals?
These molecules do not have an even number of electrons, so they are always searching for an extra electron which they can “partner” with (steal from) to become once again stable, chemically.
However, the damage doesn’t stop there, as the new molecule, say a piece of a cell wall, is now also missing an electron and has become another free radical.
This snowball effect can wreak havoc on healthy tissue.
How do Free Radicals and Antioxidants Interact?

The human body is composed of many different types of cells joined together by chemical bonds.
Numerous atoms form a molecule, and numerous molecules form a cell. When the combination of cells occurs, it forms tissue, and numerous tissues form an organ.
Atoms–>Molecules–>Cells–>Tissue–>Organs
Atoms consist of a nucleus at their center, neutrons, protons, and electrons.
Protons are positively charged particles in the atom’s nucleus. Electrons are negatively charged particles that surround the nucleus.
In Chemistry, the number of protons in the atom’s nucleus determines the number of electrons.
Electrons are involved in chemical reactions and are the substance that bonds atoms together to form molecules. Electrons orbit around an atom in varying numbers of shells depending upon the substance.
The shell closest to the nucleus is full (stable) when it has two electrons. When the first shell becomes full, the excess electrons begin to populate the second shell. When the second shell is full, excess electrons move into a third shell. This repeats until each respective shell is full and, therefore, stable.
The number of electrons in the outer shell of an atom determines its chemical behavior.
When any substance has a full outer shell of electrons, it is stable and does not usually enter into additional chemical reactions.
Atoms attempt to reach and maintain a state of maximum stability; an atom will try to fill its outer shell by:
- Gaining or losing electrons so that its outer shell has complete 8 electrons or empty it.
- Bonding with other atoms to share its own electrons with other atoms to complete its outer shell
The sharing of electrons in their outer shells with other atoms creates stability for both atoms.
When atoms share electrons, they are bound together. This satisfies both molecules’ need for maximum stability.
How Cell Damage occurs?
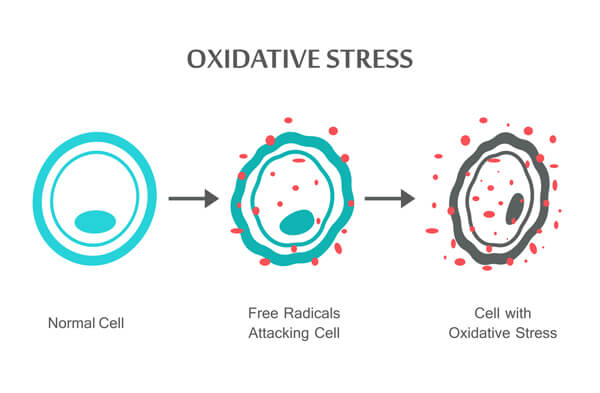
Normally, these bonds split evenly without leaving a molecule with a single, unpaired electron. However, when weaker bonds split, free radicals are formed.
This creates an unbalanced (unstable) molecule that continually attempts to restabilize itself to rebalance its shell’s electrons. It does this by “stealing” an extra electron from another nearby (stable) compound.
The first molecule now becomes stable once again, but the second molecule (which lost its electron to the first unstable molecule) becomes chemically unstable.
As this happens over and over, cell damage occurs.
Oxidation and Antioxidant
Like a banana that slowly turns brown, or when the car’s paint turns a whitish color after too much exposure to the hot sun, our bodies, too, deteriorate (become oxidated) over time due to an excessive number of free radicals.
Oxidative stress has been linked to Parkinson’s, Autism, Diabetes, Heart Disease, High Blood Pressure, Rheumatoid Arthritis, cancer, and many other diseases.
How Do Antioxidants Help To Protect The Body?
Many antioxidants such as vitamins C and E and many other nutrients are found to protect our body’s cells from these unbalanced molecules’ destructive effects.
Antioxidants have an extra electron, which they donate to free radical molecules, thus ending the constant and damaging electron-stealing process.
Antioxidants act as scavengers. In so doing, they help prevent cell damage and resultant tissue damage that can lead to diseases such as heart disease, high blood pressure, and even cancer.
What Causes Free Radicals
For a long time, free radicals gave an impression of being the big ‘enemy‘ when talking about our health, but that is actually not the case in every instance.
In truth, free radicals exist naturally inside the body. They are a product of oxidation, a process involved with respiration and other chemical reactions such as metabolism, digestion, energy production, and consumption.
The more we use oxygen, the more we are likely to produce free radicals.
In other words, just the basic actions of living life will create free radicals in your body: Breathing, Digesting food, Exercising, Sunlight – all these and more will create free radicals in your body.
In this light, we can produce more free radicals than our body can handle.
Another definition of free radicals is that they are purposefully created to neutralize viruses and bacteria, marking these common pathogens to be attacked by antibodies.
However, exposure to certain elements, including pollution, cigarette smoke, and sunlight, can also increase free radicals’ formation.
This is one more reason why smokers need to quit the habit, for the body generates about one quadrillion free radicals per stick. These elements cause damage to the tissues and delicate cell membranes.
It may also damage our DNA and disrupt our store of inherited information. Damaged cells replicate haphazardly, which is generally thought to be a precursor to cancer.
Types of Free Radicals
The most important free radicals are produced during the metabolic process, where the free radicals are derived from oxygen.
This type of free radicals is termed as Reactive Oxygen Species (ROS) or Oxygen Free Radicals (OFR).
Reactive Oxygen Species (ROS) is further classified into two groups of compounds: Radical Species and Non-Radical Species.
Radical species have at least one unpaired electron in the shells around the atomic nucleus, and they independently exist.
Non-radical species are not directly free radical, but it can easily be converted into free radicals. This is another way the free radicals are formed in the living organism.
The list of Reactive Oxygen Species (ROS) are
- Radical Species
- Superoxide
- Hydroxyl
- Alkoxyl Radical
- Peroxyl Radical
- Non Radical Species
- Hydrogen peroxide
- Singlet oxygen
- Ozone
- Organic peroxide
- Hypochlorous acid
- Hypobromous acid
Advantages and Disadvantages of Free Radicals
When the electron is stolen from an invader (e.g., bacteria, virus), the invader’s molecular structure is damaged, which leads to its death. This is a good immune function at work.
When there are no invaders left to destroy, free radicals then turn to the only other source for electrons – healthy cells.
The resulting damage to the death of healthy cells further signals the immune system for additional help. This sets up a continuous autoimmune response, which promotes chronic inflammation and tissue damage, commonly referred to as oxidative stress.
Chronic overproduction of free radicals by the immune system results in inflammatory-related diseases such as arthritis, arteriosclerosis, heart attack, Type II diabetes, lupus, MS, asthma, inflammatory bowel disease, etc.
Free Radicals and Aging Explained

Virtually every definition of free radicals states that there is the reason behind aging: the development of symptoms related to heart disease, hypertension, stroke, and cancer.
It has been claimed as the cause of so many health complaints such as headaches, dry skin, and high blood pressure, all of which are aging attributes.
Aging is directly linked with free radicals for the cause of so many major diseases, including heart disease, rheumatoid arthritis, and respiratory diseases.
Aging is an important definition of free radicals. This is because signs of free radical damage refer to the signs of aging. Heart disease is acquired with age because of the damage to the fat cells, causing them to be more “sticky” and easier to adhere to the artery walls.
High blood pressure and stroke may also result because of aging. Another sign is the dryness in the skin, causing fine lines and wrinkles.
According to the free radical theory of aging, organisms age because of the accumulation of free radicals. Over time, the damage they bring to the body is manifested through symptoms describing common health pains, aches, and discomforts.
A study conducted on rats has shown a significant increase of free radicals as the rats aged.
Inevitable free radical aging emphasizes the importance of maintaining a sufficient amount of antioxidant-rich foods in our diet.
It is important to note that free radical damage accumulates with age, just like the discoloration you see when an apple slice is left out in the air.
Free radicals and cancer
As we know, the overproduction of free radicals can lead to cell damage, and the damage may turn into cell death.
But, if cell death doesn’t occur, it may damage cell DNA, which plays a crucial role in cancer cell development.
DNA damage can lead to mutation, and this is the event where cells cannot repair the damage.
So, as per the study, due to the change in genetic material, the cell loses its control, resulting in tumor development.
Reactive oxygen species (both free radical and non-radical species) are actively involved in regulating cellular processes and direct interaction with DNA, proteins, lipids, and enzymes.
The excess production of Reactive Oxygen Species (ROS) always gives a fair chance for cancer development.
The only safest option to stop cancer development would be to stop the overproduction of free radicals.
How free radicals can cause heart disease
It’s not just the eating of fats that can cause the buildup of fat on the inner walls of arteries, but the fat cells that have been damaged by free radicals.
This is called “oxidized fat,” which causes the fat to become more “sticky,” enabling the fat to adhere to the artery walls more easily.
Some of the symptoms experienced by free radical damage
- Tremor
- Insomnia
- Headaches
- Acid reflux
- High blood pressure
- Edema
- Acne
- Eczema
- Dry skin
- Dry eyes
- Low energy & fatigue
- Allergy symptoms
- Attention Deficit Disorder
- Bedwetting
- Constipation
- Gastrointestinal problems
- Immune system problems
- Joint pain
- Migraines
- Nerve pain
- Mood swings
- Swollen lymph nodes
- Over-weight conditions
- Cysts
- Anxiety
- Poor circulation
- Cold sores
- Hearing loss
- Stroke symptoms
Most Common Diseases Linked to Free Radicals
- Cancer
- Heart Disease (Atherosclerosis)
- Stroke
- Diabetes
- Hypertension
- Cataracts
- Macular Degeneration
- Rheumatoid Arthritis
- Osteoarthritis
- Allergies
- Allergic Rhinitis
- Fibromyalgia
- Chronic Fatigue Syndrome
- Sunburns
- Adult Respiratory Distress Syndrome
- Gum Disease
- Autoimmune Diseases
- Nephritis (Kidney Disease)
- Lupus
- Premature Aging And Wrinkling
- HIV/Aids
- Congestive Heart Failure
- Asthma
- Chronic Lung Disease (Copd)
- Crohn’s Disease
- Ulcerative Colitis
- Peptic Ulcer Disease
- Parkinson’s Disease
- Sickle Cell Disease
- Skin Cancer
- Leukemia
- Lymphoma
- Alzheimer’s Dementia
- Attention Deficit Disorder
- Hepatitis (Liver Disease)
- Cerebral Trauma
- Retinopathy
- Diverticulitis/Colitis
- Cystic Fibrosis
- Melasma
- Psoriasis
- Radiation Injury
- Immunodeficiencies
- Infertility
- Menstrual Disorders
- Endometriosis
- Huntington’s Chorea
- Behcet’s Disease
- Vascular Dementia
- Peripheral Vascular Disease
How to reduce free radicals in the body?
To reduce free radicals in the body, we need antioxidants, which are found in most foods.
Antioxidants provide an extra electron, which rebalances unstable cells. As a result, it prevents free radicals from harming healthy tissue in our bodies.
Fruits and Vegetables are a good source of antioxidants needed to help fight diseases and premature aging.
According to a research study, a balanced diet requires 8 ounces (231 g) of vegetables or 18 ounces (500 g) of combined fruits and vegetables each day.
There are many foods we can consume that are very high in antioxidants. For instance, dark chocolate with unprocessed cacao has one of the highest ORAC (Oxygen radicals absorbance capacity) Values of all antioxidants.
Likewise, the fruits like blueberries, acai berries, kiwi, apples, avocados are filled with antioxidants, and the vegetables like spinach, red bell peppers, Brussel sprouts, tomatoes, and carrots are enriched with antioxidants.
Along with fruits and vegetables, you can also look for nuts like walnut, oats, and yogurt for additional benefits.
So, take a walk through the fruit and vegetable aisle in the grocery store. Choose fresh fruits and vegetables high in antioxidants and help your body reduce the oxidative damage caused by these unbalanced cells.
By eating your fruits and veggies, you will give free radicals what they want… an extra electron!
Will Antioxidants supplements help?
Many naturally occurring antioxidants like vitamins C and E, beta-carotene, glutathione can serve as an antioxidant supplement.
But an expert has really advised taking antioxidants from natural sources, not from supplements form.
Research and study have mixed reviews regarding antioxidant supplements.
A research study on the efficacy of antioxidant supplements in Prostate Cancer prevention has shown not many benefits.
Similarly, another study has shown that consumption of antioxidant supplements can low the risk of lung cancer.
Also, the study showing the long term use of beta-carotene as supplements helps in reducing age-related problems.
As per the analysis, an overdose of antioxidant supplements (beta-carotene or vitamin E) other than the recommended daily allowance (RDA) would result in complications and significantly increase the risk of dying.
The bottom line
Free radicals normally result in damage to the cells, molecules, and atoms in the body, disrupting normal cell functions, which results in an imbalance to normal body growth and functions.
To combat free radicals, we need antioxidants already produced by our body or take them from dietary sources or supplements.
The free radicals could be the early sign of the body that the cell is fighting against diseases or the formation of age-related free radicals.
The common source of age-related free radicals is processed food and an unhealthy lifestyle. A balanced diet, including super healthy foods, would give you a recommended daily dose of antioxidants.